Emphasis on pharmaceutical industry in the Union Budget 2026-27 can be observed from the fact that the Finance Minister (FM) mentioned ‘biopharma’ four times, ‘pharmacies’ once, ‘medicines’ 8 times, and ‘drugs’ 7 times in the Budget Speech. In contrast to this, the FM had mentioned the word, ‘medicines’ 11 times, ‘drugs’ 7 times, and ‘pharmaceutical’ twice in her Budget Speech of 2025-26.
Biopharma got the desired attention due to announcement of Biopharma SHAKTI (Strategy for Healthcare Advancement through Knowledge, Technology, and Innovation). ‘Shift in the disease burden’ is the rationale behind the strategy. The FM stated in her Budget Speech that India’s disease burden is observed to be shifting towards non-communicable diseases, like diabetes, cancer, and autoimmune disorders. The proportion of deaths due to Non-Communicable Diseases (NCD) in India increased to 61.8% in 2016 from 37.9% in 1990 as per estimates in a study report titled, “India: Health of the Nation’s States” – The India State-Level Disease Burden Initiative in 2017. On World Hypertension Day on May 17, 2023, the Government had unveiled the “75/25” initiative with the aim to provide standardized care to 75 million individuals living with hypertension and diabetes by December 2025.
Further, the FM stated that biologic medicines are key to longevity and quality of life at affordable costs. In this background, the FM proposed the Biopharma SHAKTI with an outlay of ₹10,000 crore over the next 5 years to develop India as a global Biopharma manufacturing hub.
This is highly desirable as by 2023 itself, 7 of the top 10 selling products were biologics. The Production-linked Incentive (PLI) Scheme for Pharmaceuticals that was approved by the Union Cabinet on February 24, 2021 with a financial outlay of ₹15,000 Crore and the production tenure from FY 2022-23 to FY 2027-28 recognized the importance of biopharma. In Category 1 of the Scheme, biopharmaceuticals were mentioned as the very first item. Autoimmune drugs, anti-cancer drugs, and anti-diabetic drugs were listed in the Category 3 of this PLI Scheme.
Biopharma SHAKTI aims at building the ecosystem for domestic production of biologics and biosimilars. A Biopharma-focused network with 3 new National Institutes of Pharmaceutical Education and Research (NIPER) and upgrading 7 existing ones, is part of the Strategy. Emphasis has been laid on creating a network of over 1,000 accredited India Clinical Trials sites. Much desired attention has been on strengthening regulatory capability as the Union Budget proposed to strengthen the Central Drugs Standard Control Organisation (CDSCO) to enable it to meet global standards and approval timeframes through a dedicated scientific review cadre and specialists.
Union Budget 2026-27 has provided relief to patients, particularly those patients who are suffering from cancer as the Basic Customs Duty (BCD) has been exempted on 17 drugs or medicines. Existing list (List 3 in the notification no. 45/2025 – Customs dated October 24, 2025) has 112 lifesaving drugs or medicines (including diagnostic test kits) listed.
Under the Union Budget 2026-27, seven (07) more rare diseases have been added for the purposes of exempting import duties on personal imports of drugs, medicines and Food for Special Medical Purposes (FSMP) used in their treatment. Existing list (List 22 in the notification no. 45/2025 – Customs dated October 24, 2025) has 51 rare diseases listed.

This reduction in the rate of Basic Customs Duty (BCD) will provide relief to the patients as the reduction in prices will improve affordability of these drugs. Innovator companies such as Novartis, Eli Lilly, AstraZeneca, AbbVie, Bayer, and Bristol Myers Squibb (BMS) are going to gain on account of this reduction in the customs duty.
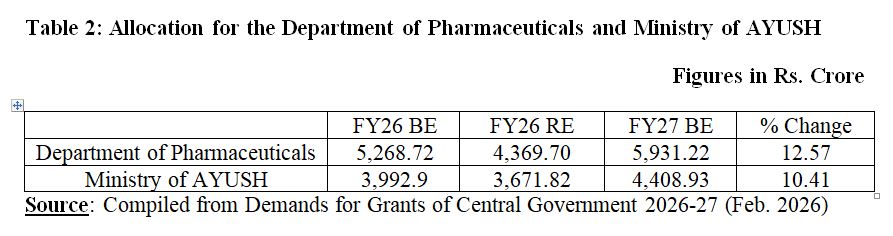
The Department of Pharmaceuticals (DoP) has been allocated ₹5,931.22 Crore for FY27. Allocation to the DoP has been increased by 12.57 per cent as FY26 BE (Budgetary Estimates) put the allocation at ₹5,268.72 Crore. Combined allocation for three Production-linked Incentive (PLI), i.e., PLI Scheme for Bulk Drugs, Medical Devices, and Pharmaceuticals for 2026-27 has been increased to ₹2,499.84 Crore which meant 2.24 per cent increase as compared to FY2025-26 as an amount of ₹2,444.93 Crore was earmarked for FY2025-26.
Union Budget 2026-27 has given significant attention to India’s traditional medicine system. AYUSH (Ayurveda, Yoga & Naturopathy, Unani, Siddha, Sowa-Rigpa, and Homeopathy) will benefit as the Union Budget has proposed setting up of 3 new All India Institutes of Ayurveda, upgradation of AYUSH pharmacies and Drug Testing Labs for higher standards of certification ecosystem, and make available more skilled personnel. The proposal to upgrade the WHO Global Traditional Medicine Centre in Jamnagar will bolster evidence-based research, training, and awareness for traditional medicine. In this regard, it is worthwhile to note that export of AYUSH (Ayurveda, Yoga & Naturopathy, Unani, Siddha, Sowa-Rigpa, and Homeopathy) and Herbal products stood at USD 688.89 million in 2024-25 by registering a growth of 6.11 per cent as export of these items stood at USD 649.2 million in 2024-25.
As highlighted by the FM in the Budget Speech that post-COVID, Ayurveda gained a global acceptance and recognition like ancient Indian yoga. Further, exporting quality Ayurvedic products helps in backward integration as it benefits farmers who grow the herbs and the youth who process the products (Budget emphasis). Recent initiatives in this direction are positive, e.g., the AYUSH Quality Mark programme was launched by the Prime Minister during the 2nd WHO Summit on Traditional Medicine (17–19 December, 2025). The Ayush Export Promotion Council (AYUSHEXCIL) which was registered as Section 8 company on January 4, 2022, has been entrusted with anchoring the Ayush Quality Mark programme. Formal recognition for AYUSH has been received in bilateral trade agreements, e.g., the India–Oman Comprehensive Economic Partnership Agreement (CEPA) and the India–New Zealand Free Trade Agreement (FTA). These agreements have provision for dedicated annexes on health-related services and traditional medicine.
The proposal regarding Hubs for Medical Value Tourism will benefit AYUSH too as these Hubs will serve as integrated healthcare complexes that combine medical, educational and research facilities. These Hubs will have AYUSH Centres, Medical Value Tourism Facilitation Centres and infrastructure for diagnostics, post-care, and rehabilitation as proposed in the Union Budget. Overall, the Scheme encompasses the support to States in establishing Regional Medical Hubs, in partnership with the private sector, for promoting India as a hub for medical tourism services.
To conclude, Biopharma SHAKTI is likely to help in positioning India as a global biologics/biobetters/biosimilars manufacturing hub, Customs Duty reduction will make eligible drugs affordable, increased allocation for AYUSH systems will promote these traditional systems while encouraging exports, and increase in combined allocation for PLI Schemes (Pharma/Medical Devices/Bulk Drugs) will add to domestic production capacity.
Dr. Anil Kumar Angrish- Associate Professor (Finance and Accounting) and In-Charge, Department of Pharmaceutical Management, NIPER S.A.S. Nagar (Mohali), Punjab
Disclaimer: Views are personal and do not represent the views of the Institute.






