During this week, a headline on India and China talks regarding Reverse-Trade Model for Drugs and Pharmaceuticals attracted eyeballs. The move was considered a favourable development by Indian Pharmaceutical Industry. The Pharmaceutical Exports Promotion Council (Pharmexcil) is optimistic that it can unlock about $6 Bn in pharmaceutical exports as against exports of Active Pharmaceutical Ingredients (APIs) from China to India.
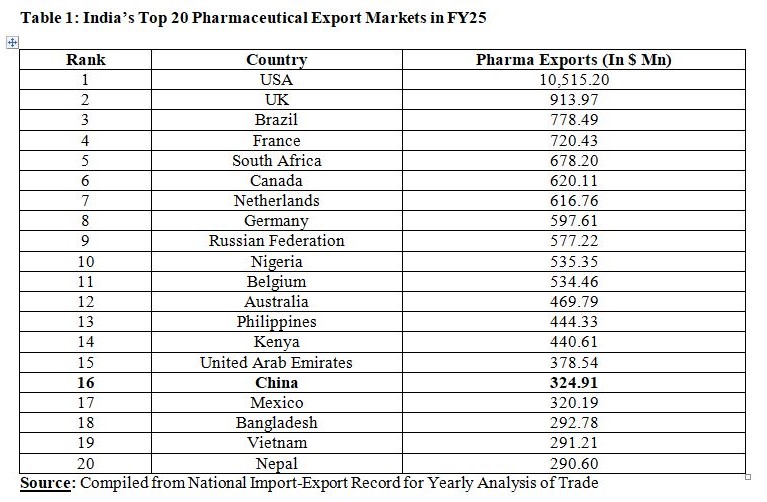
This is a positive development for Indian Pharmaceutical Industry as India is highly dependent on the US market and there is urgent need to diversify geographic reach due to tariff moves of Trump administration. India exported Drugs and Pharmaceuticals worth US$30.38 Bn in FY25. The USA remained the number one market for Indian Drugs and Pharmaceuticals. In comparison to exports of Drugs and Pharmaceuticals worth $10,515.2 Mn to the USA, exports to China stood at just $324.91 Mn. In other words, export of drugs and pharmaceuticals to China formed just 3.09 per cent of the pharmaceutical exports to the USA. Export of pharmaceutical products to China worth $324.91 Mn seems insignificant when seen in the background that China is the second most populous country after India, and the country is also the world’s second largest economy by nominal Gross Domestic Product (GDP), just next to the USA.
Pharmaceutical Industry of China is ranked as the 2nd largest in the world. At the same time, China has the largest number of people living with diabetes worldwide (The Lancet, Dec. 2024). In 2023, China became the country with the largest number of Alzheimer’s patients in the world. China’s National Health Commission (NHC) statistics reveal that China’s population aged 60 and above had reached 310 million by the end of 2024. It is almost 22.0 per cent of the Total Population of China. These numbers show that there is immense scope for Indian Pharmaceutical Industry in China.
In 2nd week of November 2025, four Indian Pharma firms (Hetero Labs, Cipla, Natco Pharma, and Annora Pharma) were in news as these companies won bids under the Volume-based Procurement (VBP) bidding process conducted by the Chinese Health ministry. Cipla and Hetero Labs won the bid to supply Dapagliflozin tablets which are used in treatment of diabetes. Kunshan Rotam Reddy Pharmaceutical Co., a Chinese subsidiary of Dr. Reddy’s Laboratories (DRL) had also won the bid. Out of 55 drugs from different therapeutic categories such as anti-infectives, anti-tumour treatments, allergy treatments, etc., Indian pharma firms secured contracts to supply seven drugs. Indian companies had edge due to low pricing as otherwise, the market is dominated by Pharma MNCs and Chinese players.
In the 2nd week of November 2025 itself, Zydus Lifesciences also received approval from National Medical Products Administration (NMPA) of China (Venlafaxine Extended-Release Capsules indicated for treatment of Major Depressive Disorder, Generalized Anxiety Disorder, Social Anxiety Disorder and Panic Disorder). It became the first approval for the Zydus Group from NMPA. In the same week, Glenmark Pharma also got approval for one of their nasal spray (indicated for treatment of allergic rhinitis in adults and children).
It is pertinent to mention that India has trade deficit of $54.4 Bn with mainland China for April-September 2025, up from $49.6 Bn in the corresponding period of 2024. In 2024-25, India’s trade deficit stood at $99.2 Bn which used to be $1.1 Bn in 2003-04. From time to time, India had flagged its concern over the ballooning trade deficit along with non-trade barriers. In the backdrop of trade deficit, Indian pharma companies getting the orders for 7 out of 55 drugs is a positive development.
In recent past, Indian pharmaceutical companies have made serious attempts to enter in Chinese market. On July 16, 2025, Sun Pharma incorporated Sun Pharma (Hainan) Company a wholly-owned subsidiary in China. Sun Pharma announced that this subsidiary will enable Sun Pharma to establish a manufacturing base for select products through a strategic partner, aimed at facilitating their commercialisation in China.
Indian pharmaceutical companies are collaborating with Chinese companies too, e.g., in December 2024, Mankind Pharma entered into an agreement with Innovent Biologics of China to sell anti-cancer drug (Sintilimab) in India. The drug was co-developed by Eli Lilly and Innovent. Innovent will take care of manufacturing and supply of the drug, and Mankind Pharma got the exclusive rights to register, import, market, sell and distribute the drug in India.
Positive developments need to be seen in the background of other important moves in this sector. In February 2025, prices of many Key APIs were substantially lowered (40-50%) by Chinese manufacturers. This price cut was applicable for most of the 41 identified products which fall under the Production-linked Incentive (PLI) Scheme for promotion of domestic manufacturing of critical Key Starting Materials (KSMs)/Drug Intermediates (DIs) and Active Pharmaceutical Ingredients (APIs) in India. Indian manufacturers were compelled to lower the prices of those APIs to confront Chinese manufacturers. Such moves have implications for the success of PLI Schemes.
In the first-half (H1) of FY26, i.e. April to September 2025, India imported bulk drugs and intermediates to the tune of $2.24 Bn as against $2.24 Bn imports for April to September 2024. In ₹ terms, imports for H1 of FY26 stood at ₹19,335.70 Crore. In this context, it is worthwhile to note that about 72.0 per cent of imports, in value terms, were from China. In Quantity terms, import of bulk drugs and intermediates for H1 of FY26 stood at 2,51,818 MT, up from 2,45,532 MT in the same period of FY25.
Sceptics see approvals for many Indian pharmaceutical firms in the Chinese market as “too much, too fast” in the background of past ordeals. On the Chinese visit of Indian PM Narendra Modi in August 2025, the first such visit to China in seven years, China’s President Xi Jinping had told that China and India should be partners, not rivals.
Recent developments reveal that tariffs can be used as economic weapons to protect domestic industries, to fund government operations, and even coerce foreign countries into compliance. The U.S. President had stated his intention to increase the amount of tax raised by the government. Besides this, other stated objectives were: to reduce the U.S. trade deficit, to encourage consumers to buy more U.S.-made goods, and to boost investments in the U.S. Along the same lines, India should not allow pharmaceuticals to be used as next trade weapon by any economy.
Dr. Anil Kumar Angrish- Associate Professor (Finance and Accounting), Department of Pharmaceutical Management, NIPER S.A.S. Nagar (Mohali), Punjab
Disclaimer: Views are personal and do not represent the views of the Institute.





